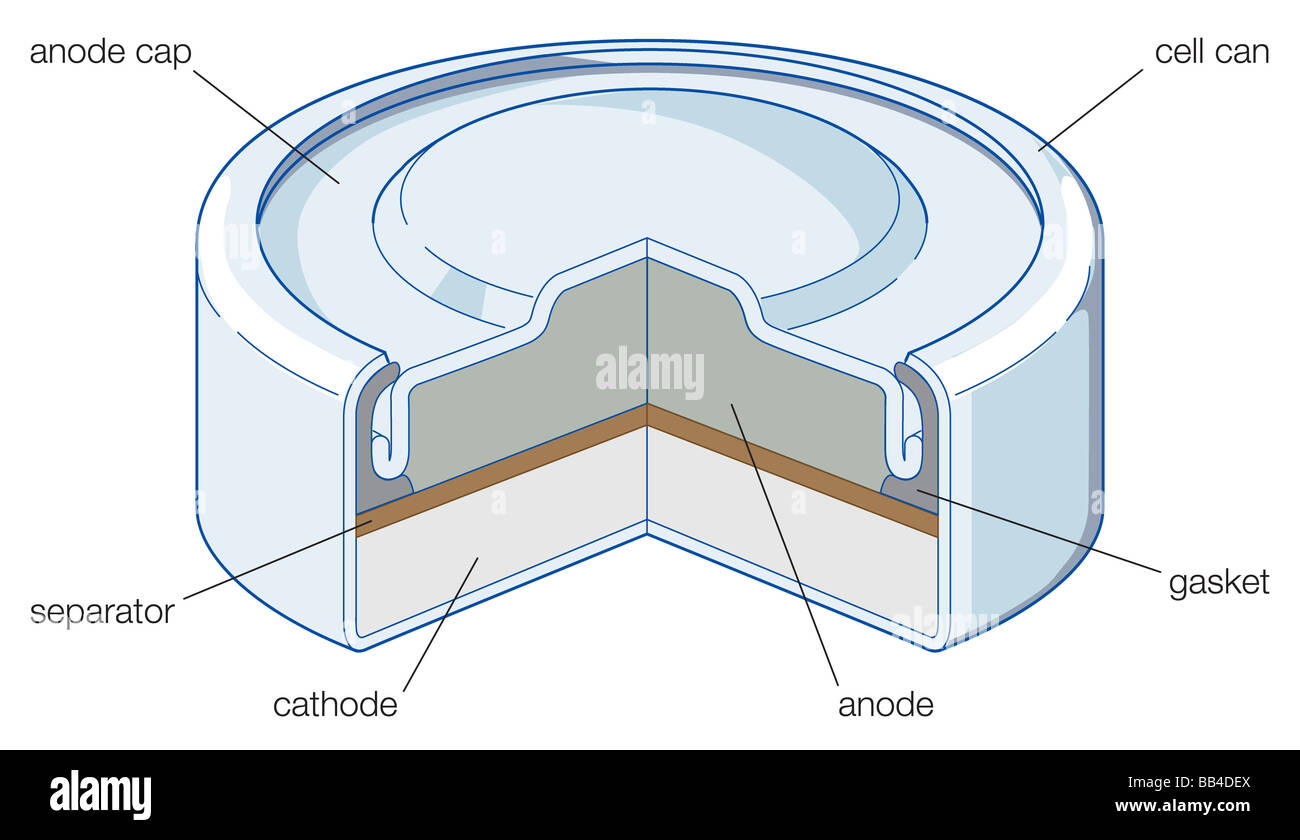
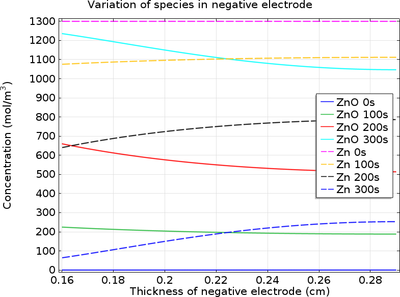
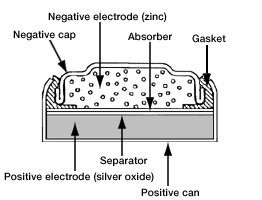
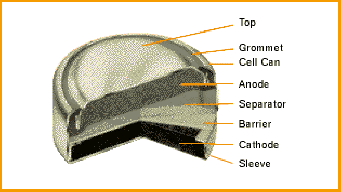
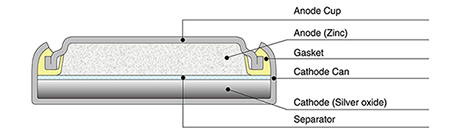
Zinc/silver oxide cell is used in hearing aids and electric watches. The following reactions occur : Zn(s) to Zn^(2+)(aq)+2e^(-) , E_(Zn^(2+)//Zn)^(@)=-0.76V Ag_(2)O+H_(2)O+2e^(-) to2Ag+2OH^(-),E_(Ag^(+)//Ag)^(@)=0.344" V" Calculate (i) Standard ...

Primary Cells A primary cell is a cell that can be used once only and cannot be recharged. The reactants cannot be regenerated. - ppt video online download

SOLVED:0.80 V For question (4), what does "X" represent? a) Tin b) Lead Cobalt Silver The net reaction of the mercury cell is Zn Hgo Hzo Zn(OH)2 Hg What substance is oxidized
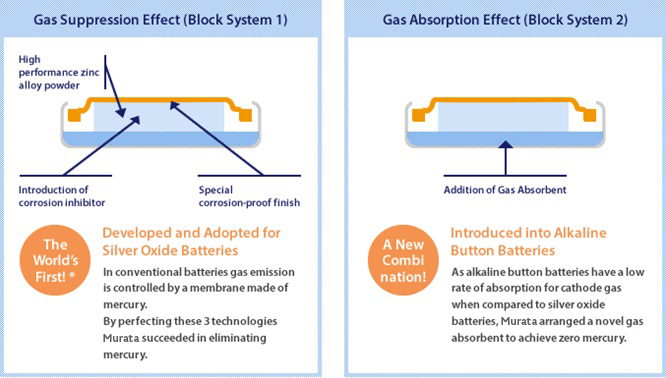
The First in the World to Achieve Silver Oxide/Alkaline Button Batteries with 0% Mercury | Tohoku Murata Manufacturing Co., Ltd.